Biochemistry Publications
Proteolytic Amyloid Digestion Assay for Fibril Polymorphisms
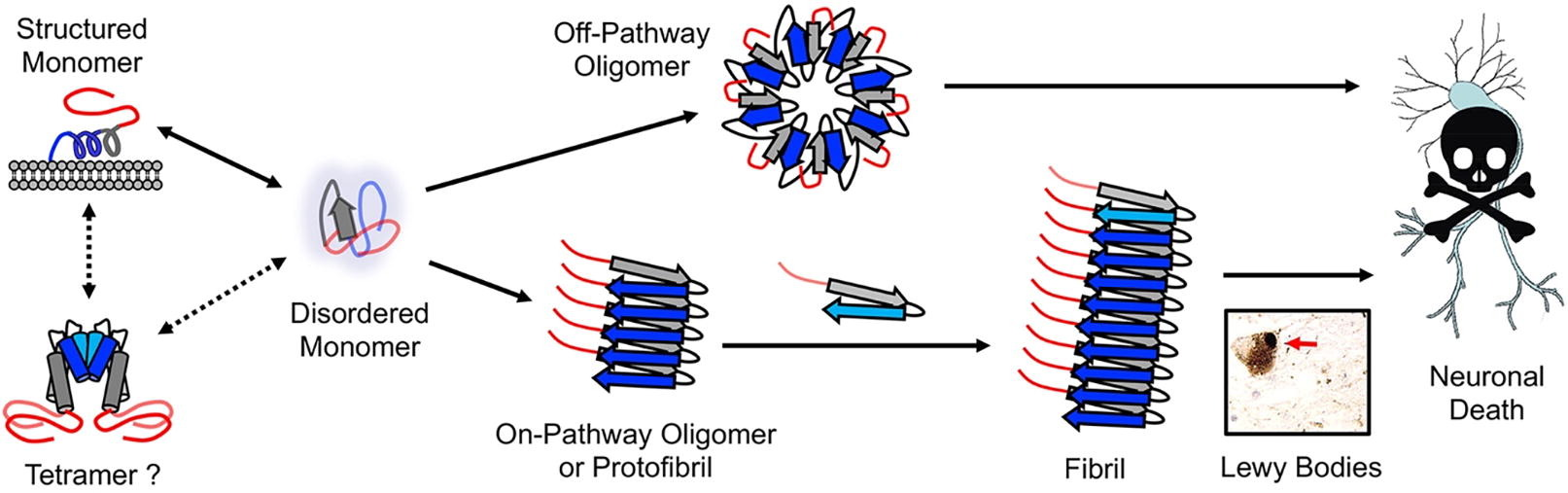
Developed a novel proteolytic digestion assay with mass spectrometry readout for rapidly distinguishing different α-synuclein fibril polymorphisms. This technique enables faster and more reproducible characterization of disease-relevant fibril structures, improving the diagnosis of Parkinsonian diseases.
Selective Radioligand Binding to α-Synuclein in MSA Tissue
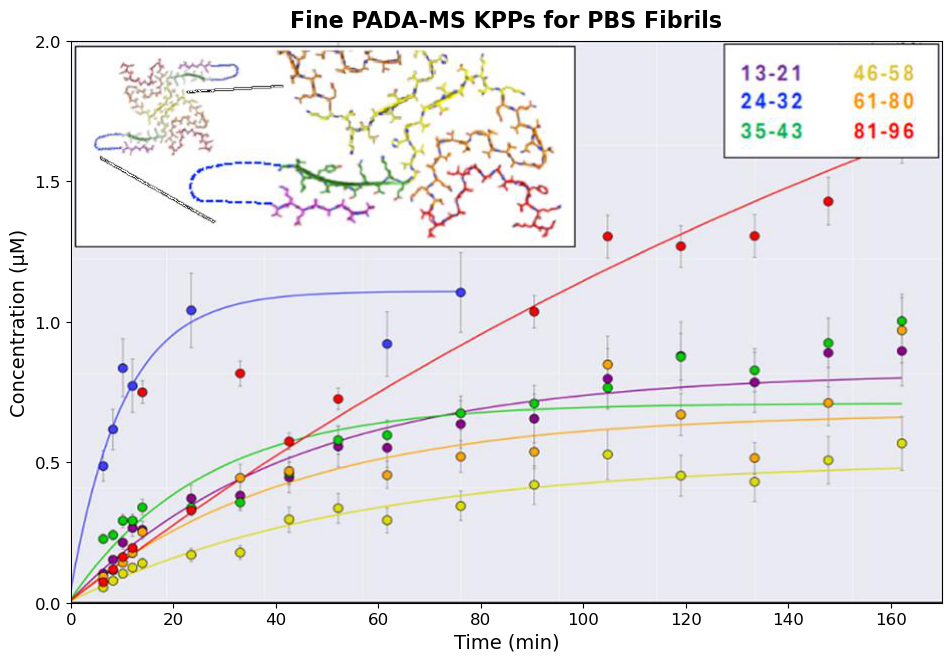
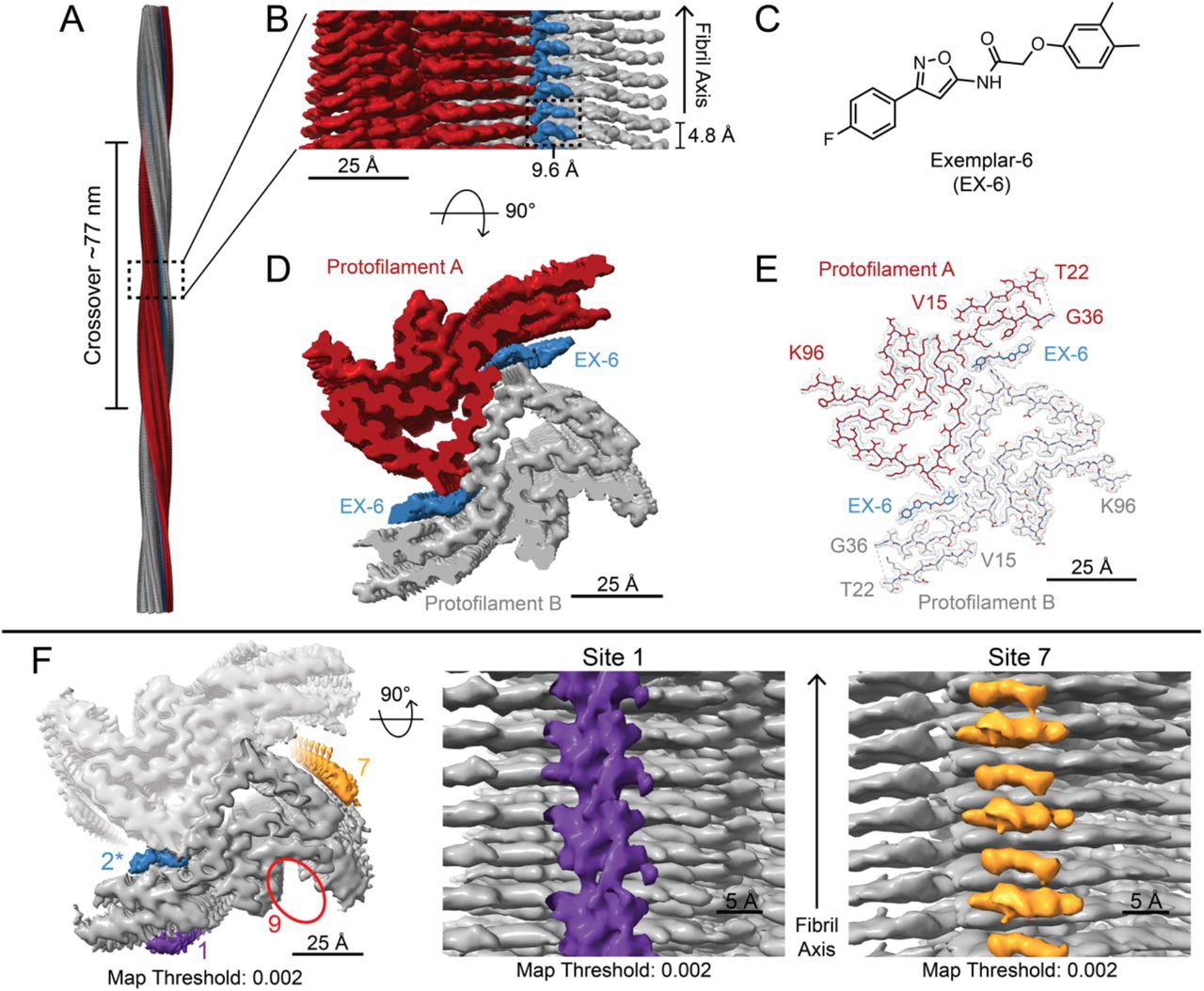
Used complementary structural and chemical biology techniques including cryo-EM, crosslinking mass spectrometry, FRET, and radioligand binding assays to characterize how the PET imaging candidate EX-6 binds to α-synuclein fibrils. Identified a specific binding pocket conserved in MSA patient-derived structures, demonstrating 10-fold selectivity for MSA over PD tissue.
Radiotracer Competition Binding with α-Synuclein Fibrils
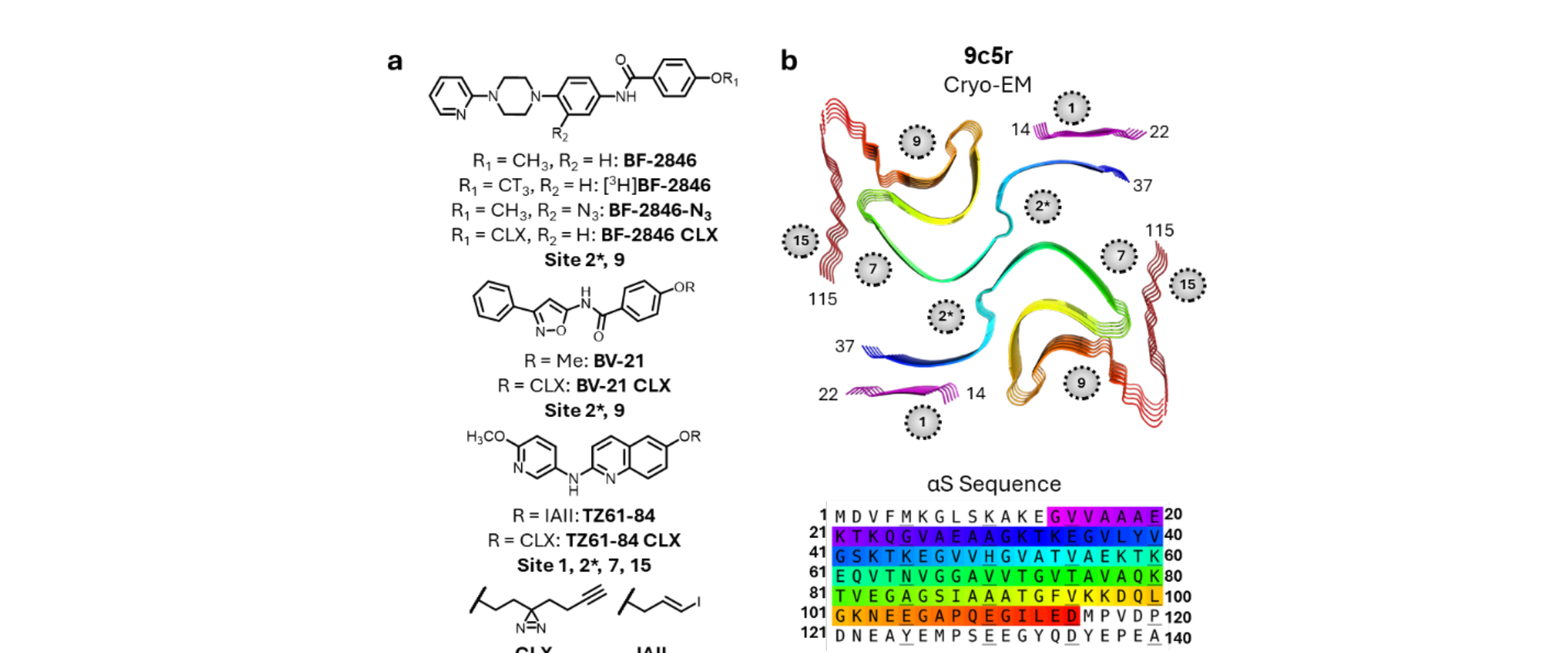
Developed a radiotracer competition binding platform to systematically evaluate how ~300 small molecules interact with α-synuclein fibrils, revealing structure-activity relationships for ligand specificity. Identified key design principles for developing selective PET imaging probes for different synucleinopathies.
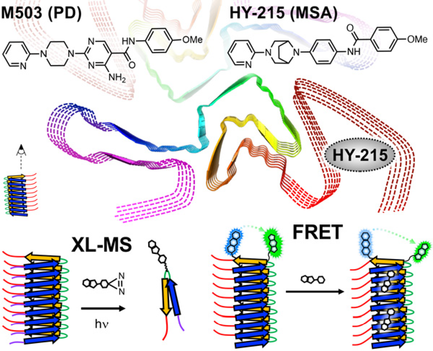
PET Probe Interactions with the C-Terminus of α-Synuclein Fibrils
Investigated how two PET imaging probe candidates (M503 and HY-215) interact with α-synuclein fibrils at the molecular level. Using photo-crosslinking mass spectrometry and FRET, discovered that the MSA-selective probe HY-215 uniquely interacts with the disordered C-terminus, highlighting the importance of often-ignored disordered fibril regions for selective diagnostics.
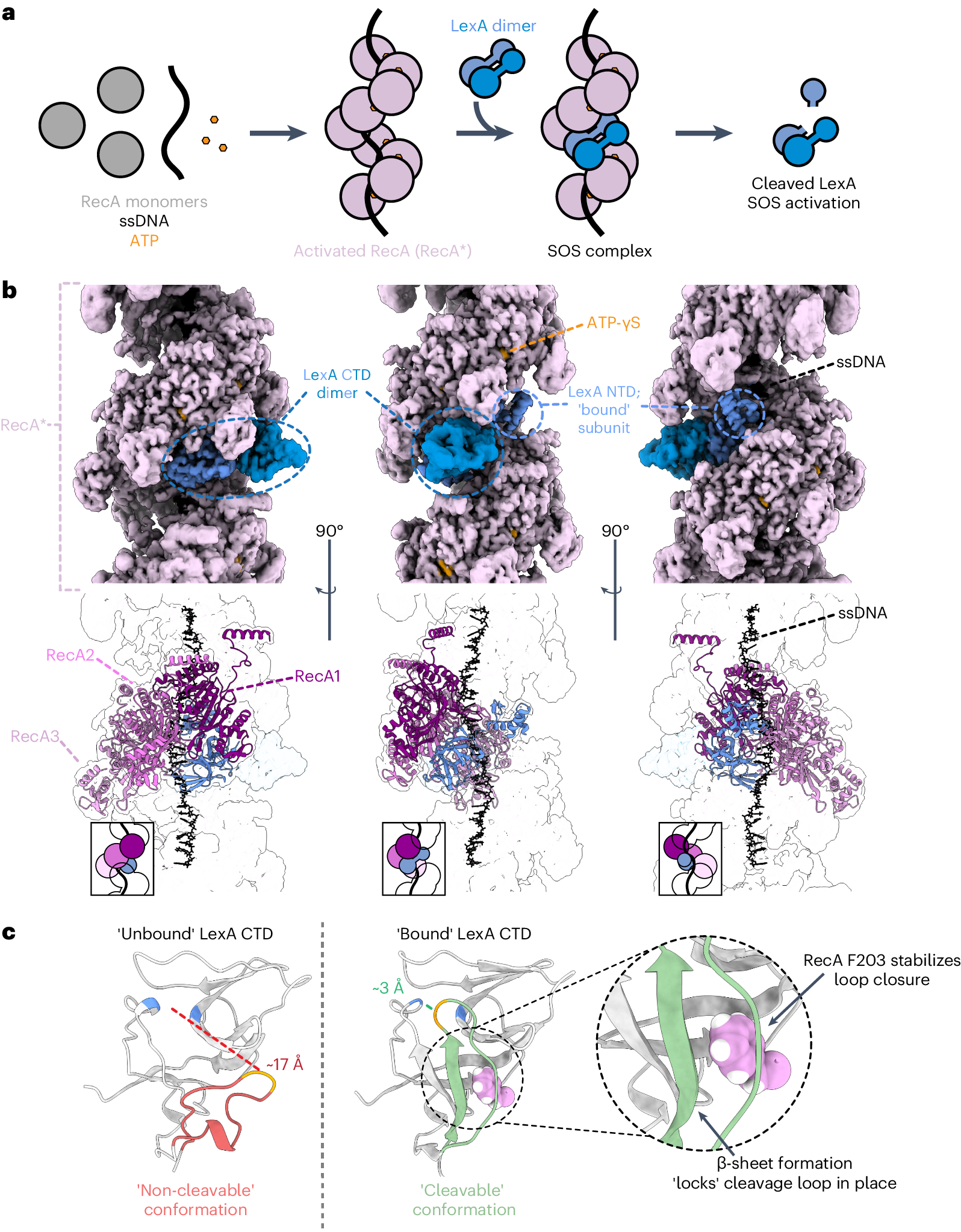
LexA-RecA* Lock-and-Key Mechanism for SOS Activation
Contributed to solving the first cryo-EM structure of the complete E. coli SOS signaling complex — full-length LexA bound to activated RecA*. The structure reveals an extensive binding interface where a single RecA residue acts as a molecular key that inserts into an allosteric pocket to trigger LexA self-cleavage, providing a foundation for therapeutics to slow antibiotic resistance evolution.
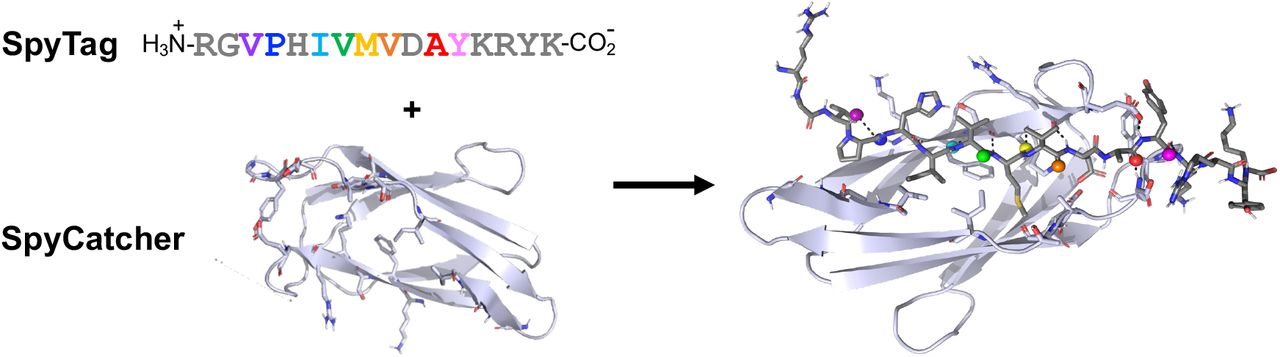
SpyCatcher Ligation Kinetics by SpyTag Thioamide Substitution
Explored how thioamide substitutions in SpyTag peptides modulate the kinetics of the SpyCatcher-SpyTag ligation reaction, a widely used bioconjugation tool. Demonstrates how subtle backbone chemical modifications can tune protein ligation rates, expanding the toolkit for controlled bioconjugation chemistry.
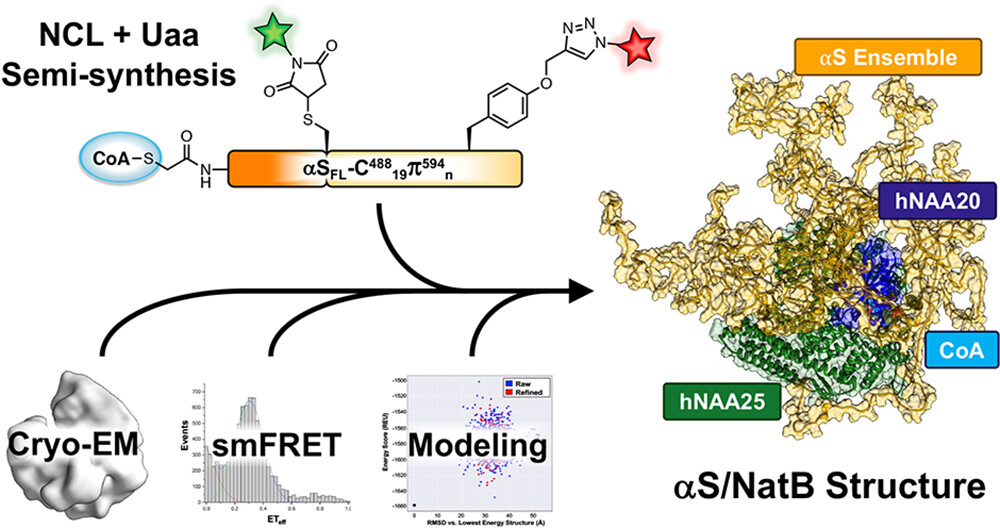
Semi-Synthetic CoA-α-Synuclein Traps NatB Acetyltransferase
First semi-synthesis of a bisubstrate inhibitor of N-terminal acetyltransferase NatB, consisting of coenzyme A linked to full-length α-synuclein via native chemical ligation. Cryo-EM and single-molecule FRET revealed that α-synuclein remains largely disordered when bound to NatB with its C-terminus expanding — providing structural insights into this key post-translational modification linked to Parkinson's disease.
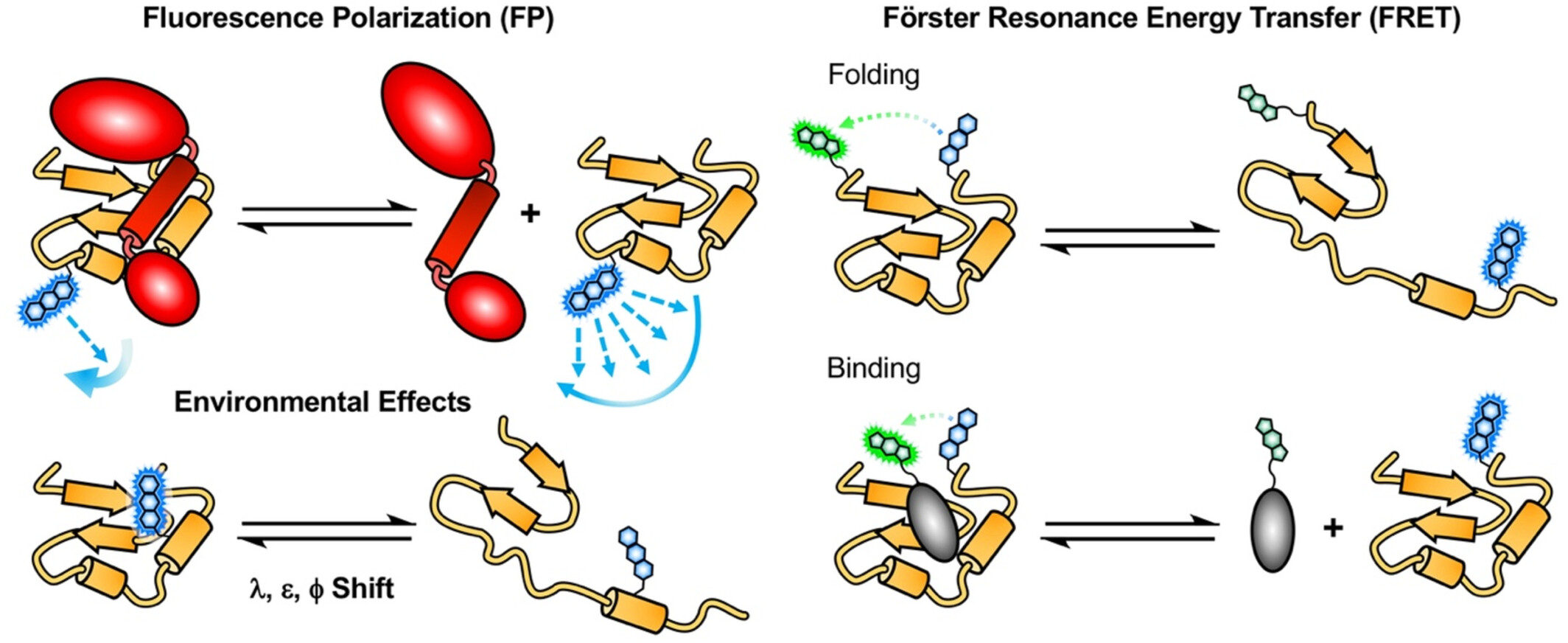
FRET with Genetically Encoded Fluorescent Amino Acids
Presents case studies using the fluorescent amino acid acridonylalanine (Acd) as a genetically encoded FRET probe to measure protein conformational changes and distances. Demonstrates how this minimally perturbing probe enables quantitative estimation of protein structure and dynamics in systems including α-synuclein and the LexA-RecA complex.
α-Synuclein Aggregation: Mutations & Post-Translational Modifications
Comprehensive study of how disease-associated mutations and post-translational modifications affect α-synuclein in vitro aggregation. Systematically evaluates the effects of familial mutations, phosphorylation, acetylation, and other modifications on fibril formation, providing important context for understanding Parkinson's disease pathogenesis.